【人気ダウンロード!】 p xylene structure 303243-P-xylene compound structure
Impurities in pXylene on Stabilwax Styrene on Stabilwax Ethylbenzene on Stabilwax TriplePhase SPME Arrow vs TriplePhase SPME Fiber Residual Solvents #1 on Rxi624Sil MS Residual Solvents and Gases on Rxi624Sil MS via HSSyringeGCFID NJ Low Level TO15 75 Component Mix on RtxVMS (60 m, 40 °C start)The two authors, WP, a US industrial chemist with more than 30 years' experience of oxidation processes, and MP, a UK academic green chemist, were first brought together by the oxidation of p‐xylene in supercritical water, which forms the final section of this chapter Their aim is to bring this story of this process to a wider chemical(need structure) Expert Answer 100% (13 ratings)
P Xylene
P-xylene compound structure
P-xylene compound structure-The two most stable resonance structures of Acridine are equivalent to each other, and both contain a pyridinelike structural unit Write a structural formula for AcridineQNitration of 1, 4dimethylbenzene (pxylene)Nitration of 1, 4dimethylbenzene (pxylene) gives a single product having the molecular formula C8H9NO2 in high yieldM, o, and pXylene are the three isomers of xylene;



P Xylene D10 D 99atom 1 Sigma Aldrich
PXylene C6H4(CH3)2 or C8H10 CID 7809 structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety/hazards/toxicity information, supplier lists, and moreSee the answer How many isomer are form by nitration of mxylene?Commercial or mixed xylene usually contains about 4065% mxylene and up to % each of o and pxylene and ethylbenzene (1) Mixed xylenes are colorless liquids that are practically insoluble in water and have a sweet odor (1) The odor threshold for mxylene is 11 ppm (4)
PXylene C8H10 structure 🎓Molecular Formula C8H10 Average mass Da Density 09±01 g/cm3 Boiling Point 1396±100 °C at 760 mmHg Flash Point 272±00 °C Molar1,4xylene C24H30 CID structure, chemical names, physical and chemical properties, classification, patents, literaturePxylene C 8 H 10 13 133 0861 002 0074 026 100 15 1 M = miscible 2 The values for relative polarity are normalized from measurements of solvent shifts of absorption spectra and were extracted from Christian Reichardt, Solvents and Solvent Effects in Organic Chemistry, WileyVCH Publishers, 3rd ed, 03
This material contains pXylene (CAS# , 99%),which is subject to the reporting requirements of Section 313 of SARA Title III and 40 CFR Part 373 Clean Air Act CAS# is listed as a hazardous air pollutant (HAP) This material does not contain any Class 1 Ozone depletorsTerephthalic acid was produced by oxidation of pxylene with dilute nitric acid Air oxidation of pxylene gives ptoluic acid, which resists further airoxidation Conversion of ptoluic acid to methyl ptoluate (CH 3 C 6 H 4 CO 2 CH 3) opens the way for further oxidation to monomethyl terephthalate, which is further esterified to dimethyl(need structure) how many isomer are form by nitration of p xylene?



P Xylene Molecular Image Photo Free Trial Bigstock


9 P Xylol Stock Illustrations Vectors Clipart Dreamstime
PXylene is an isomer xylene mainly used in the polyester industry as a raw material for synthesizing 5,6terephthalic acid Application pXylene may be used in the synthesis of ethyl 2,5dimethylphenylhydroxy malonate by reacting with ethyl oxomalonate in the presence of anhydrous stannic chloride Packaging(need structure) how many isomer are form by nitration of p xylene?Xylene is an aromatic hydrocarbon with a single ring and two methyl groups attached to this ring in positions 12, 13 and 14, called oxylene, mxylene, and pxylene, respectively


P Xylylene Glycol C8h10o2 Chemspider
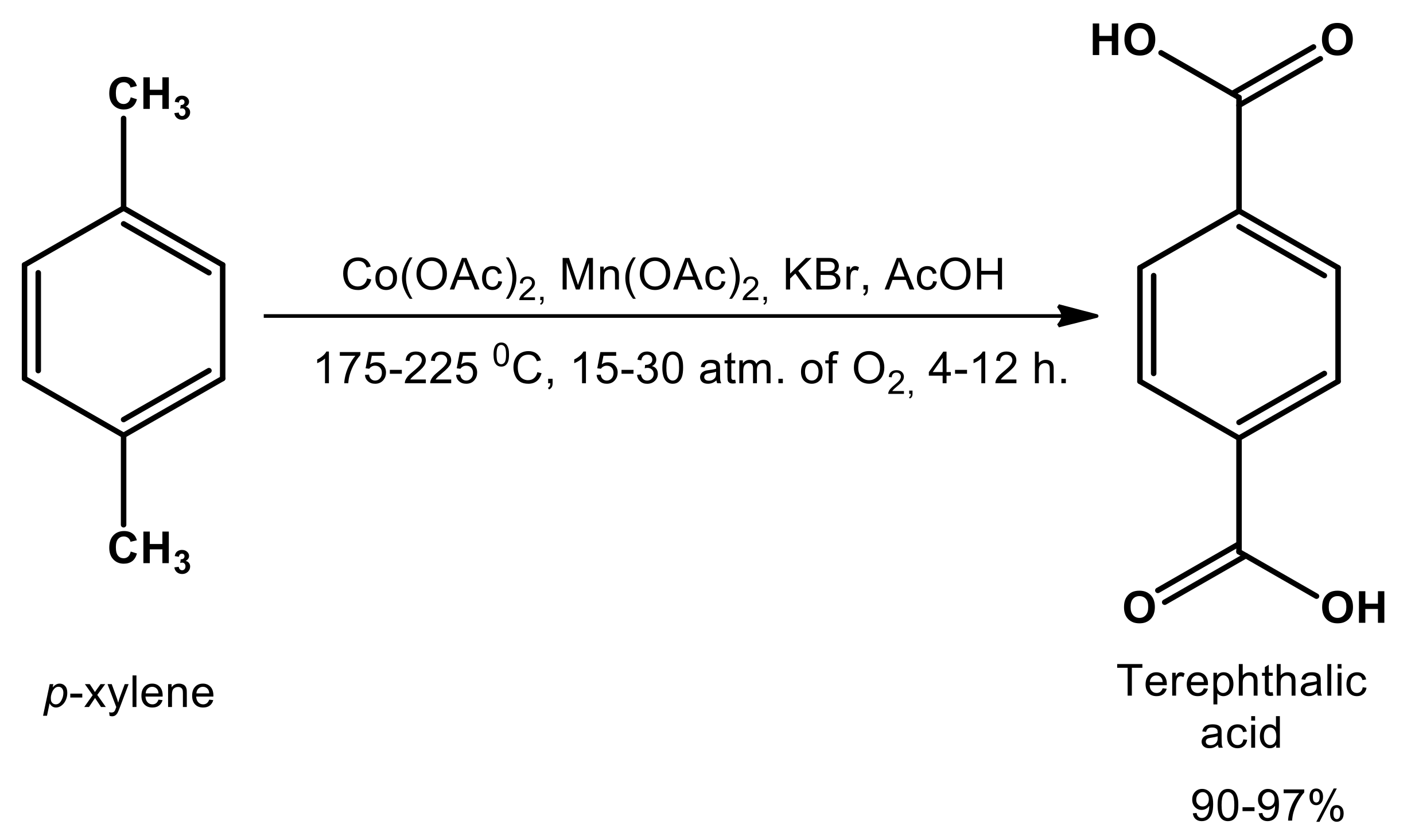


Catalysts Free Full Text Manganese And Cobalt Doped Hierarchical Mesoporous Halloysite Based Catalysts For Selective Oxidation Of P Xylene To Terephthalic Acid Html
2Chloropxylene C8H9Cl CID 328 structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety/hazards/toxicity information, supplier lists, and more COVID19 is an emerging, rapidly evolving situation Public health information (CDC) Research information (NIH)Heat capacity, c p 1870 J/(mol K) at 25°C Gas properties Std enthalpy change of formation, Δ f H o gas 190 kJ/mol Standard molar entropy, S o gas 3536 J/(mol K) Heat capacity, c p 1325 J/(mol K) at 25°C van der Waals' constants a = 3038 L 2 kPa/mol 2 b = liter per molePXylene is an aromatic hydrocarbon It is one of the three isomers of dimethylbenzene known collectively as xylenes The p stands for para, indicating that the two methyl groups in pxylene occupy the diametrically opposite substituent positions 1 and 4 It is in the positions of the two methyl groups, their arene substitution pattern, that it differs from the other isomers, oxylene and mxylene All have the same chemical formula C6H4(CH3)2 All xylene isomers are colorless and highly flamm


Di Chloro P Xylene Cas 79 4 Chemsrc
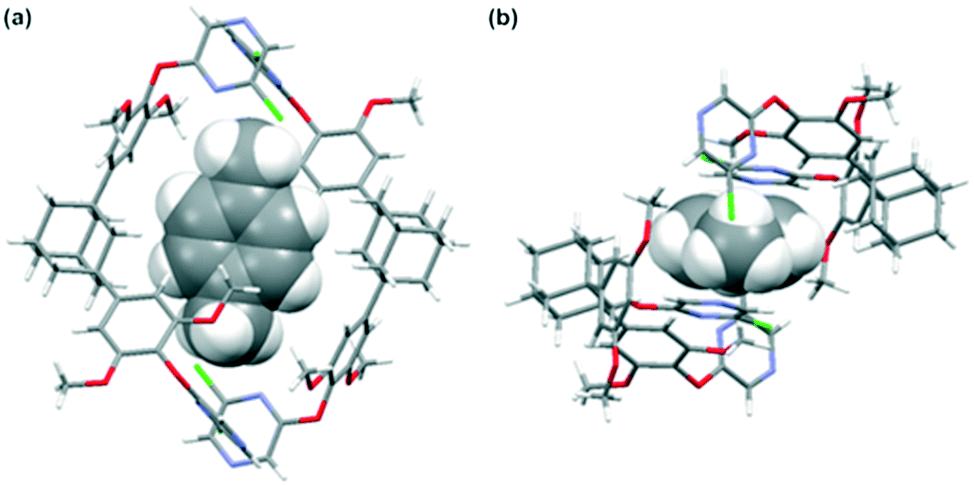


Separation Of P Xylene From Aromatic Compounds Through Specific Inclusion By Acyclic Host Molecule Crystengcomm Rsc Publishing
Search results for p xylene at SigmaAldrich Compare Products Select up to 4 products *Please select more than one item to compare1,4dimethylbenzene ( pxylene) Permanent link for thisChemsrc provides pXylene(CAS#) MSDS, density, melting point, boiling point, structure, formula, molecular weight etc Articles of pXylene are included as well


Q Tbn And9gcravnnhb1ldj2s0rhmfe5ldt6j32eu6j5rozppxdes7hxl4p5yk Usqp Cau


P Xylene
There are three xylenes mxylene (CASR# 10), oxylene (CASR# ), pxylene (CASR# ), since their molecular formula is the same they are called isomers Their chemical structures are slightly different Since they are often sold and used as 'mixed isomers', unless a particular isomer is specifically mentioned the informationThis structure is also available as a 2d Mol file or as a computed 3d SD file The 3d structure may be viewed using Java or Javascript Other names mXylene;Read "Structure of (trifluoromethylbenzene)(p‐xylene)chromium(0) at 163 K, Acta Crystallographica Section C Structural Chemistry" on DeepDyve, the largest online rental service for scholarly research with thousands of academic publications available at your fingertips
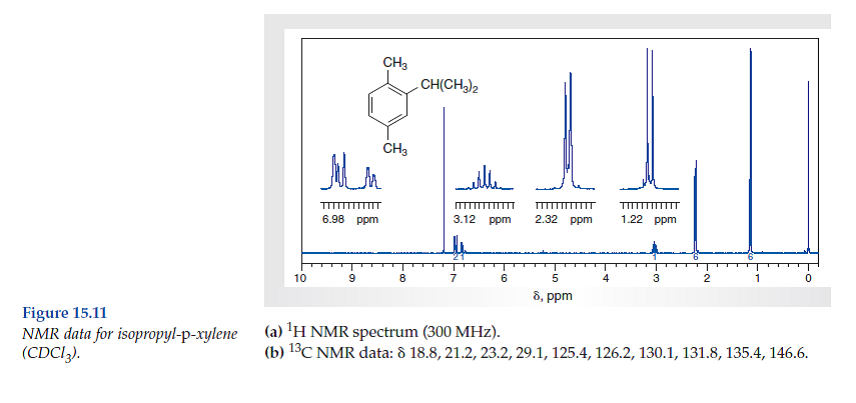


Solved Consider The Spectral Data For Isopropyl P Xylene Figs Chegg Com


Btx Chemistry Wikipedia
(need Structure) This problem has been solved!The thermodynamic stability of mxylene over oxylene or pxylene can be deduced by considering the hyperconjugative effectMore specifically, resonance forms with a positive charge on methylsubstituted carbons are more important than resonance forms with a negative charge on them, as the methyl group stabilises the positively charged carbon by hyperconjugationXylene, xylol or dimethylbenzene is any one of three isomers of dimethylbenzene, or a combination thereof With the formula (CH3)2C6H4, each of the three compounds has a central benzene ring with two methyl groups attached at substituents They are all colorless, flammable liquids, some of which are of great industrial value The mixture is referred to as both xylene and, more precisely, xylenes


P Xylene



Oxidation Of P Xylene To Terephthalic Acid And 4 Carboxybenzaldehyde Download Scientific Diagram
SigmaAldrich offers a number of p Xylene products View information & documentation regarding p Xylene, including CAS, MSDS & morePXylene is an isomer xylene mainly used in the polyester industry as a raw material for synthesizing 5,6terephthalic acid Application pXylene may be used in the synthesis of ethyl 2,5dimethylphenylhydroxy malonate by reacting with ethyl oxomalonate in the presence of anhydrous stannic chloride PackagingThe 3d structure may be viewed using Java or Javascript Isotopologues 1,4Di(methyld3)benzened4;


The Chemical Structure Of O Xylene M Xylene P Xylene And Ethylbenzene Download Scientific Diagram


2h10 P Xylene C8h10 Pubchem
The structure of pxylene was similarly refined using the 180 K Xray single crystal structure of van Konigsveld et al as the starting model Profile fits to the diffraction data from both compounds are shown in Fig 2, Fig 3and the refined structural parameters given in Table 1, Table 2 Download Download highres image (145KB) Download Download fullsize image1 Introduction pXylene (PX), an important largevolume commodity chemical in the petrochemical industry, is mainly used to produce terephthalic acid (TPA) , , which is the comonomer in a condensation with monoethylene glycol (MEG) for the manufacturing of polyethylene terephthalate (PET) , Over 50 million tons of PET are globally produced per year , and PET is widely used in common1,3dimethylbenzene (mxylene) Permanent link for this species



Draw The Structure Of P Xylene Bis Triphenylphosphonium Nitrosodisulfonate Ph3pch2c6h4ch2pph3 On So3 2 And Indicate The Likely Position Of The Unpaired Electron S Homeworklib
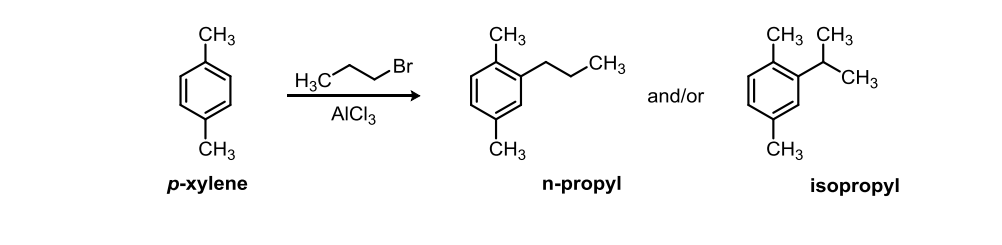


Solved A From This Friedel Crafts Alkylation Of P Xylene Chegg Com
Paraxylene (PXylene) Paraxylene (PXylene) Paraxylene, also called pxylene, is a chemical essential to the process of manufacturing PET plastic bottles and polyester fiber Share Follow Us on Facebook Twitter YouTube RSS Print this Page Uses & Benefits Safety InformationThis material contains pXylene (CAS# , 99%),which is subject to the reporting requirements of Section 313 of SARA Title III and 40 CFR Part 373 Clean Air Act CAS# is listed as a hazardous air pollutant (HAP) This material does not contain any Class 1 Ozone depletorsThe two authors, WP, a US industrial chemist with more than 30 years' experience of oxidation processes, and MP, a UK academic green chemist, were first brought together by the oxidation of p‐xylene in supercritical water, which forms the final section of this chapter Their aim is to bring this story of this process to a wider chemical



P Xylene 99 Pure Acros Organics Fisher Scientific


O Xylene 95 47 6
(need structure) Expert Answer 100% (13 ratings)Xylene Structure Let us see the xylene molecular structure now Image will be Uploaded Soon Xylene exists in three different isomeric forms These isomers can be distinguished by the designations ortho (o), meta (m) and para (p), that specify to which carbon atoms (of the benzene ring) the two methyl groups are attachedThe 2D chemical structure image of PXYLENE is also called skeletal formula, which is the standard notation for organic molecules The carbon atoms in the chemical structure of PXYLENE are implied to be located at the corner(s) and hydrogen atoms attached to carbon atoms are not indicated – each carbon atom is considered to be associated with enough hydrogen atoms to provide the carbon atom
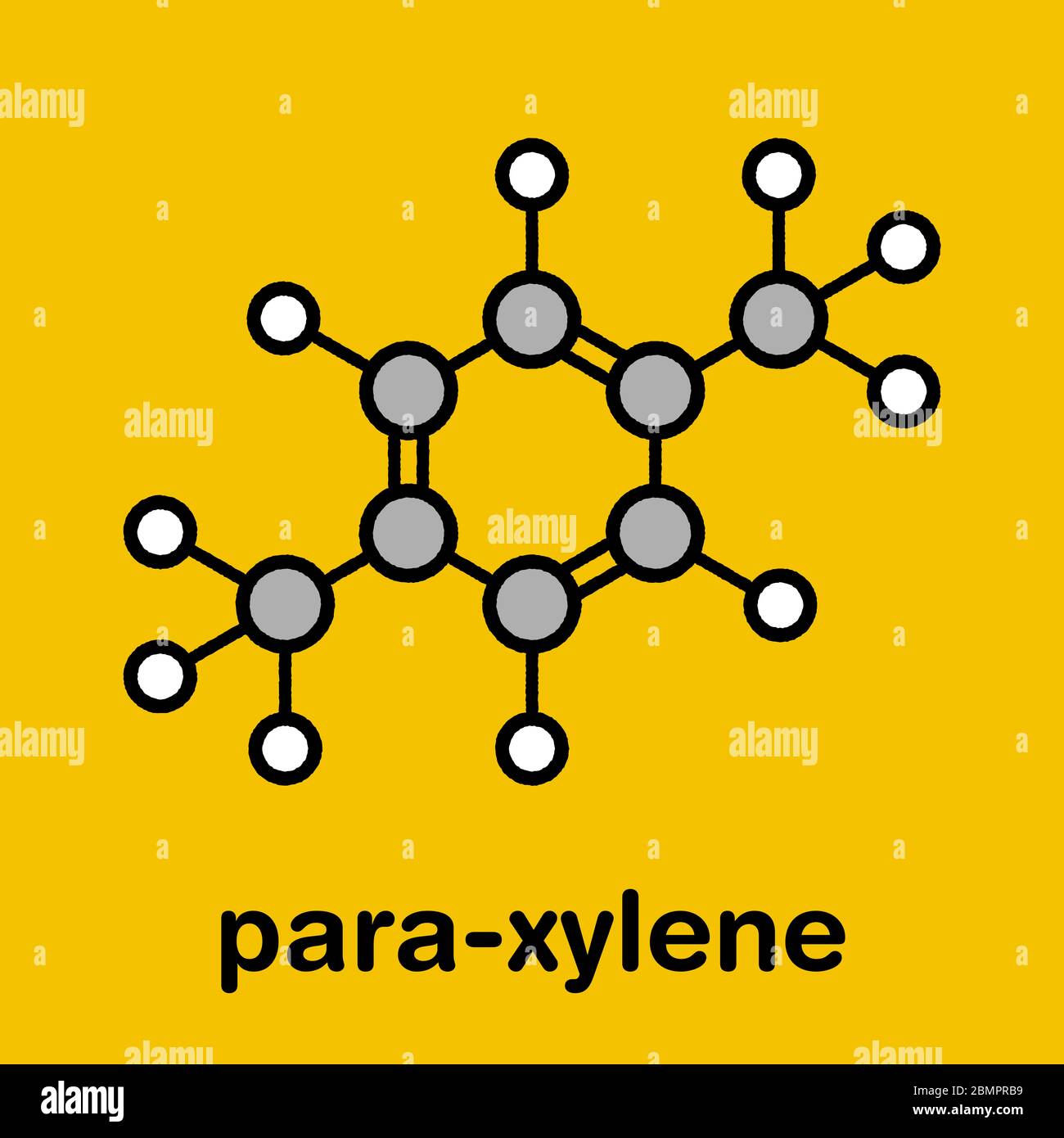


Para Xylene P Xylene Aromatic Hydrocarbon Molecule Stylized Skeletal Formula Chemical Structure Atoms Are Shown As Color Coded Circles Hydrogen White Carbon Grey Stock Photo Alamy
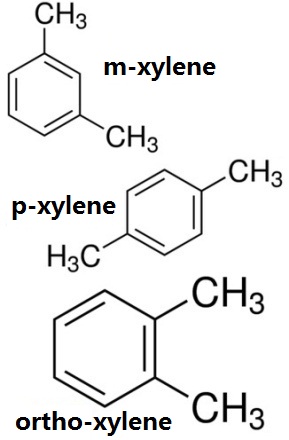


O Xylene 95 47 6
(need Structure) This problem has been solved!Chemsrc provides pXylene(CAS#) MSDS, density, melting point, boiling point, structure, formula, molecular weight etc Articles of pXylene are included as wellCommercial or mixed xylene usually contains about 4065% mxylene and up to % each of oxylene and pxylene and ethylbenzeneXylenes are released into the atmosphere as fugitive emissions from industrial sources, from auto exhaust, and through volatilization from their use as solvents



2 3 5 6 Tetrafluoro P Xylene Structure C8h6f4 Over 100 Million Chemical Compounds Mol Instincts
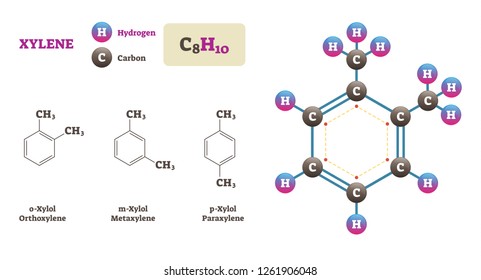


Xylene Hd Stock Images Shutterstock
The thermodynamic stability of mxylene over oxylene or pxylene can be deduced by considering the hyperconjugative effectMore specifically, resonance forms with a positive charge on methylsubstituted carbons are more important than resonance forms with a negative charge on them, as the methyl group stabilises the positively charged carbon by hyperconjugationXylene, any of three isomeric dimethylbenzenes which have the same chemical formula, C 6 H 4 (CH 3) 2, but different molecular structure, used as solvents, as components of aviation fuel, and as raw materials for the manufacture of dyes, fibres, and filmsThe three isomers, designated ortho (o), meta (m), and para (p), differ structurally only in the location of the methyl groupsSynonym αCyanopxylene, 4Methylbenzyl cyanide, 4Methylphenylacetonitrile, pTolylacetonitrile Linear Formula CH 3 C 6 H 4 CH 2 CN Molecular Weight



T Butyl P Xylene Structure C12h18 Over 100 Million Chemical Compounds Mol Instincts
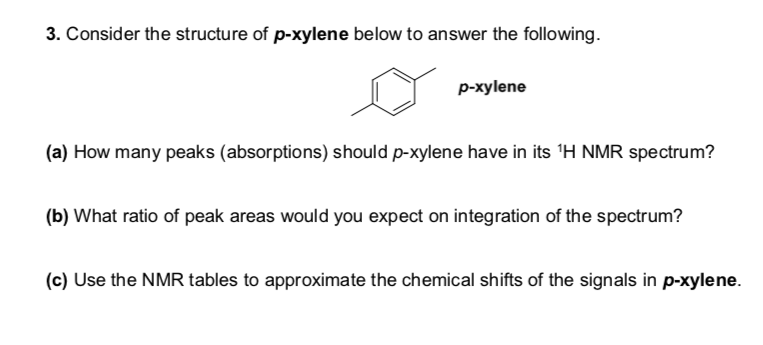


Solved 3 Consider The Structure Of P Xylene Below To Ans Chegg Com
MXylene (metaxylene) is an aromatic hydrocarbonIt is one of the three isomers of dimethylbenzene known collectively as xylenesThe mstands for meta, indicating that the two methyl groups in mxylene occupy positions 1 and 3 on a benzene ringIt is in the positions of the two methyl groups, their arene substitution pattern, that it differs from the other isomers, oxylene and pxyleneXylene is a colorless mixture of chemicallyrelated hydrocarbons that often finds use as a solvent for paints and printing inks It's very good at dissolving compounds that dissolve poorly in water, which is why it is so useful Its ability to do so stems from its properties and its chemical structureSearch results for p xylene at SigmaAldrich Compare Products Select up to 4 products *Please select more than one item to compare



P Xylene Anhydrous 99 106 42 3 Sigma Aldrich


Xylene High Resolution Stock Photography And Images Alamy
Xylene Structure Let us see the xylene molecular structure now Image will be Uploaded Soon Xylene exists in three different isomeric forms These isomers can be distinguished by the designations ortho (o), meta (m) and para (p), that specify to which carbon atoms (of the benzene ring) the two methyl groups are attached(need Structure)how Many Isomer Are Form By Nitration Of P Xylene?Structure, properties, spectra, suppliers and links for 2bromopxylene,



Figure 1 From The Crystal Structure And Morphology Of 2 4 6 8 10 12 Hexanitro 2 4 6 8 10 12 Hexaazaisowurtzitane Cl P Xylene Solvate A Joint Experimental And Simulation Study Semantic Scholar



P Xylene Structure C8h10 Over 100 Million Chemical Compounds Mol Instincts
(need Structure)how Many Isomer Are Form By Nitration Of P Xylene?Structure, properties, spectra, suppliers and links for pXylene, , , ,Xylene, any of three isomeric dimethylbenzenes which have the same chemical formula, C 6 H 4 (CH 3) 2, but different molecular structure, used as solvents, as components of aviation fuel, and as raw materials for the manufacture of dyes, fibres, and films The three isomers, designated ortho ( o ), meta ( m ), and para ( p ), differ structurally only in the location of the methyl groups


P Xylene Ftir Spectrum Spectrabase



Selective P Xylene Production From Biomass Derived Dimethylfuran And Ethylene Over Zeolite Beta Nanosponge Catalysts Sciencedirect
1 Introduction pXylene (PX), an important largevolume commodity chemical in the petrochemical industry, is mainly used to produce terephthalic acid (TPA) , , which is the comonomer in a condensation with monoethylene glycol (MEG) for the manufacturing of polyethylene terephthalate (PET) , Over 50 million tons of PET are globally produced per year , and PET is widely used in commonOther names Benzene, 1,4dimethyl;Structure, properties, spectra, suppliers and links for Xylene,


2 1 Phenylethyl P Xylene C16h18 Pubchem



Fluorescence And Photophysical Properties Of Xylene Isomers In Water With Experimental And Theoretical Approaches Royal Society Open Science
PXylene C8H10 structure 🎓Molecular Formula C8H10 Average mass Da Density 09±01 g/cm3 Boiling Point 1396±100 °C at 760 mmHg Flash Point 272±00 °C Molar118 °C TCI D0605 °C Alfa Aesar °C Merck Millipore 4135, 8416 119 °C JeanClaude Bradley Open Melting Point Dataset 534 118 °C JeanClaude Bradley Open Melting Point Dataset 1175 °C JeanClaude Bradley Open Melting Point Dataset °C Alfa Aesar A 1181 °C LabNetwork LN °C SynQuest 1175 °C Parchem – fineParaxylene (PXylene) Paraxylene (PXylene) Paraxylene, also called pxylene, is a chemical essential to the process of manufacturing PET plastic bottles and polyester fiber Share Follow Us on Facebook Twitter YouTube RSS Print this Page Uses & Benefits Safety Information
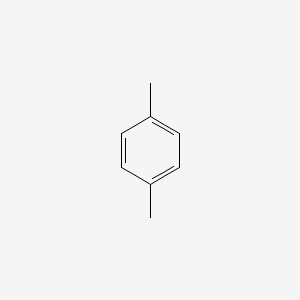


P Xylene C6h4 Ch3 2 Pubchem


P Xylene
See the answer How many isomer are form by nitration of mxylene?
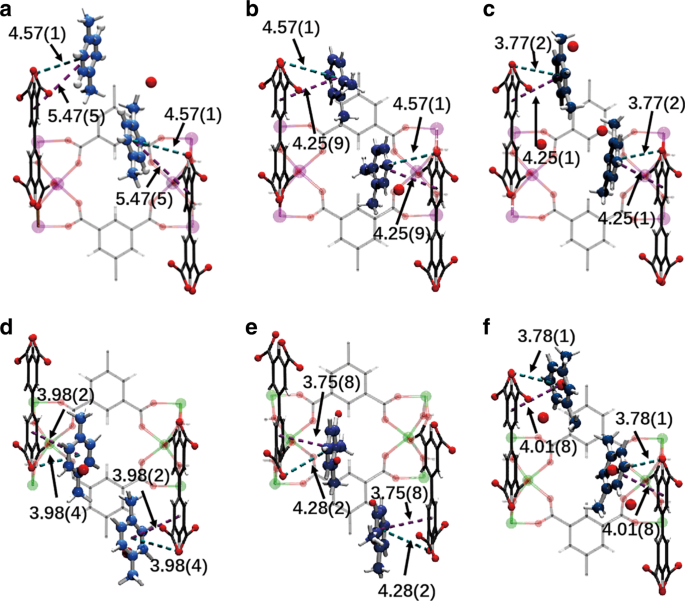


Refinement Of Pore Size At Sub Angstrom Precision In Robust Metal Organic Frameworks For Separation Of Xylenes Nature Communications



Ozonolysis Of P Xylene Gives


P Xylene 106 42 3 7w Gk1927 Cymit Quimica S L


Xylene Hd Stock Images Shutterstock



File P Xylene Png Wikimedia Commons


Similar To P Xylene Chemical Safety Models Suppliers Regulation And Patents



P Xylene 106 42 3 C8h10 Density Melting Point Boiling Point Structural Formula Synthesis



Chemical Structure Of Deuteriated P Xylene And The Principal Axis Download Scientific Diagram
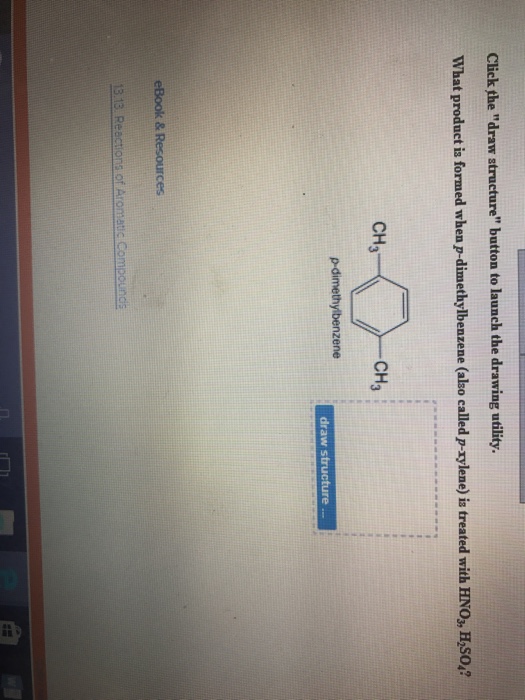


Solved What Product Is Formed When P Dimethylbenzene Als Chegg Com
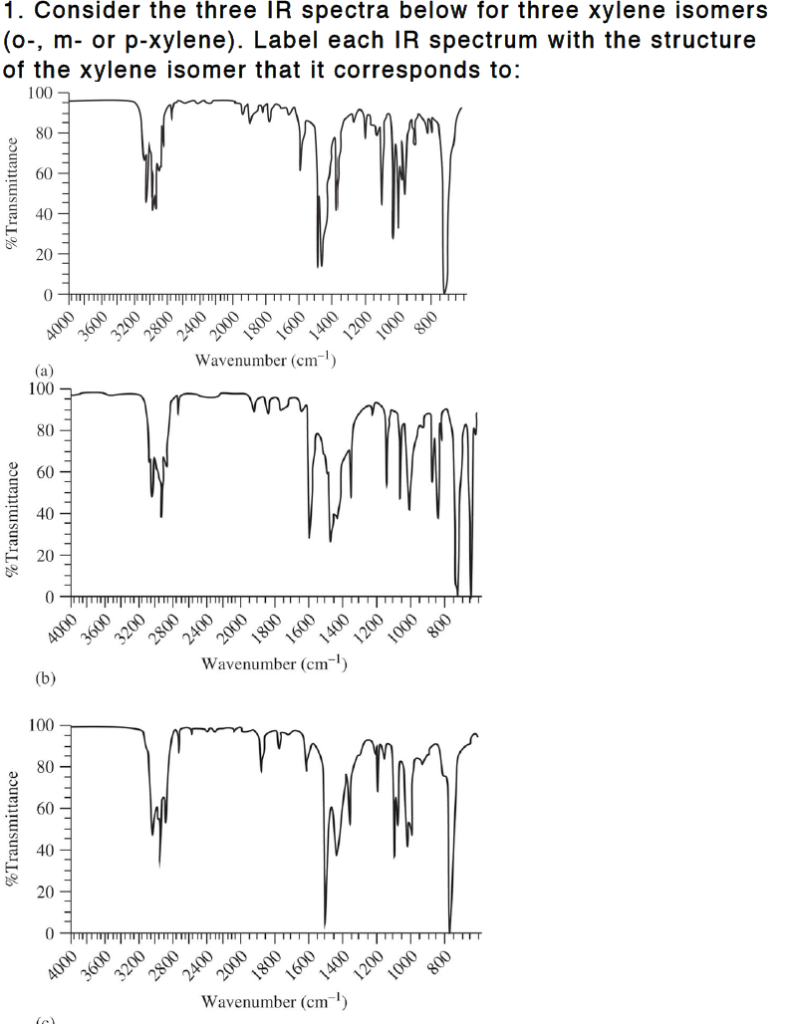


Solved 1 Consider The Three Ir Spectra Below For Three X Chegg Com
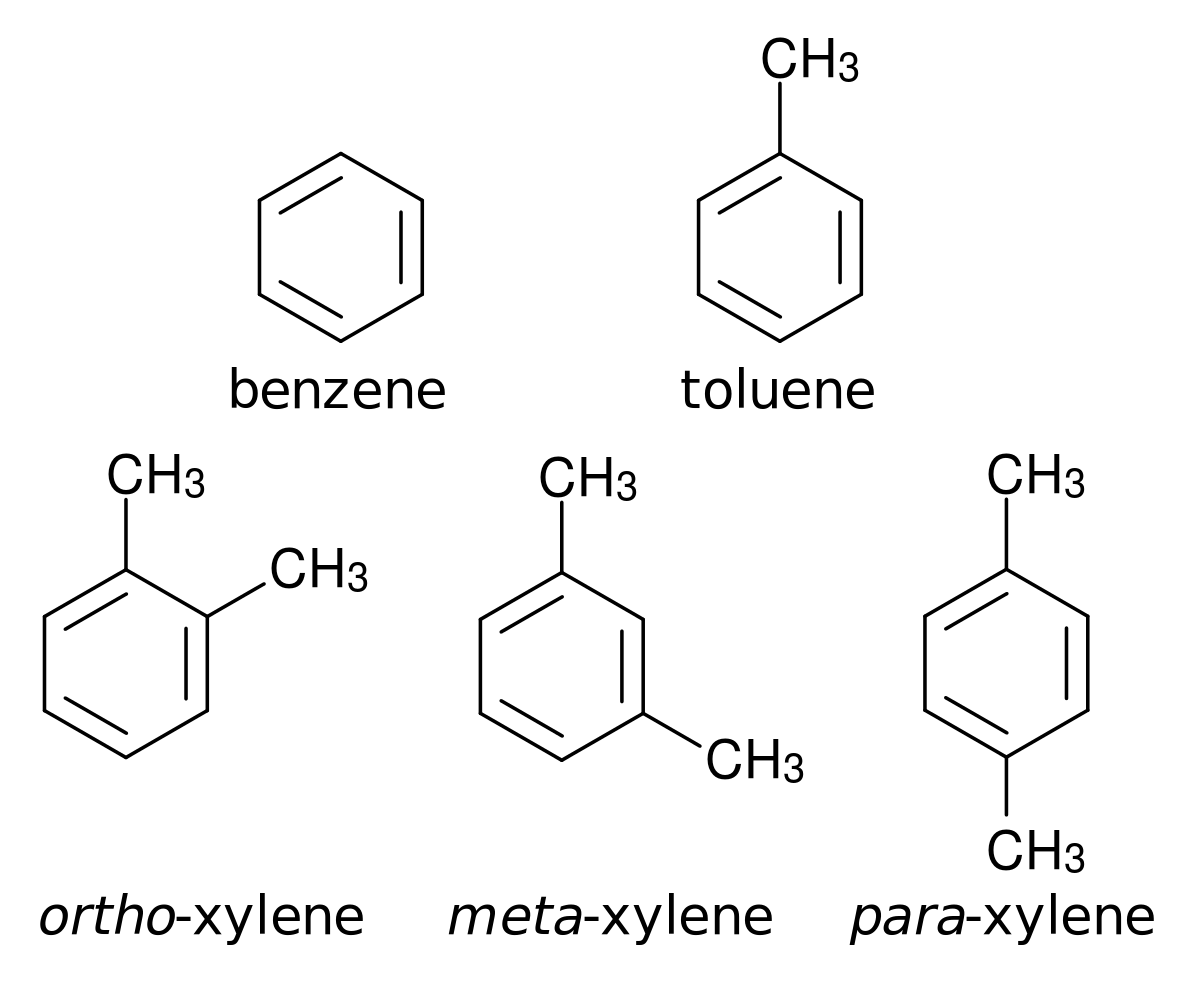


Btx Chemistry Wikipedia



2 Iodo P Xylene Formula C8h9i Over 100 Million Chemical Compounds Mol Instincts



Alpha Chloro P Xylene 98 Acros Organics Fisher Scientific



2 3 6 Tribromo P Xylene 80 2 Wiki
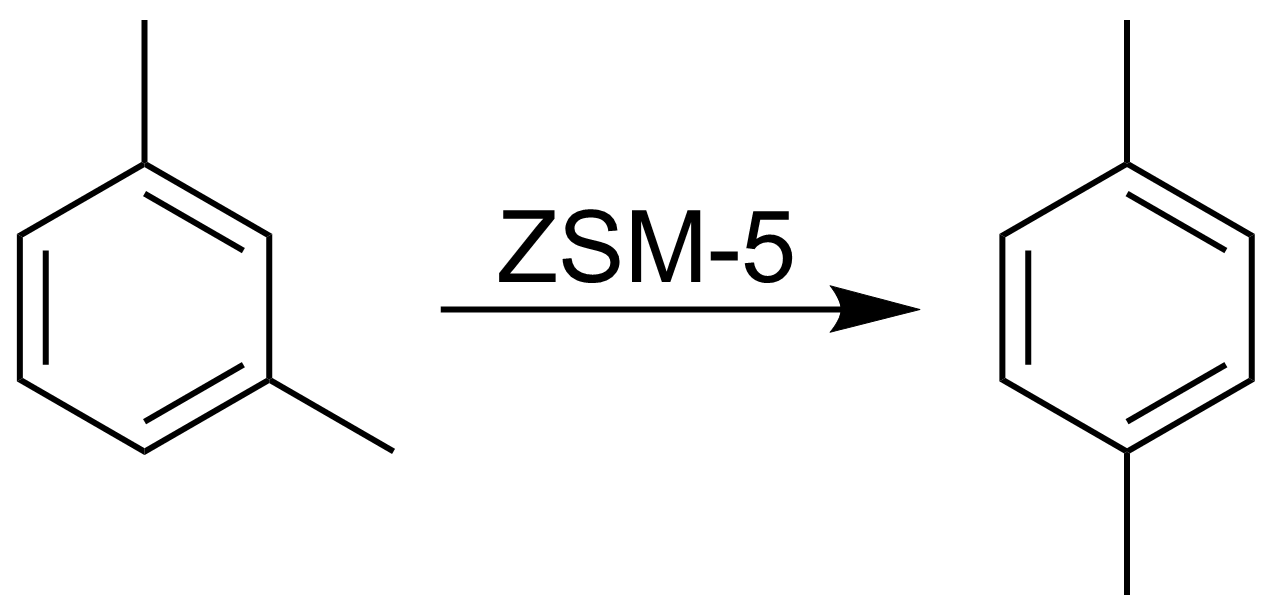


File M Xylene To P Xylene Png Wikipedia



File P Xylene 0 Svg Wikimedia Commons



P Xylene Molecular Image Photo Free Trial Bigstock



Pxylene Molecular Structure Isolated On White Stock Photo Download Image Now Istock



Separation Of Para Xylene And Meta Xylene By Extraction Process Using Aqueous Cyclodextrins Solution Sciencedirect



Solved Click The Draw Structure Button To Launch The Dr Chegg Com



Pxylene Molecular Structure Isolated On White Stock Photo Download Image Now Istock



The Chemical Structure Of O Xylene M Xylene P Xylene And Ethylbenzene Download Scientific Diagram



A Brief Review Of Para Xylene Oxidation To Terephthalic Acid As A Model Of Primary C H Bond Activation Sciencedirect



Tetrachloro P Xylene Structure C8h6cl4 Over 100 Million Chemical Compounds Mol Instincts



Pxylene Molecular Structure Isolated On White Stock Photo Download Image Now Istock
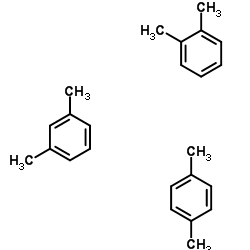


M Xylene O Xylene P Xylene Cas 03 3 Chemsrc



2 Bromo 1 4 Dimethylbenzene 99 553 94 6 Sigma Aldrich
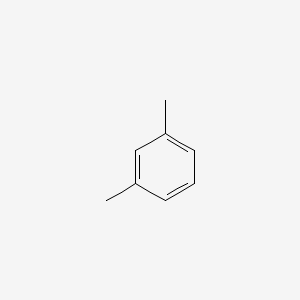


M Xylene C6h4 Ch3 2 Pubchem
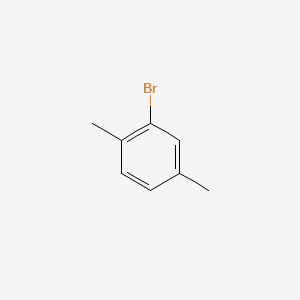


2 Bromo P Xylene C8h9br Pubchem


1 4 2 Bromo P Xylene
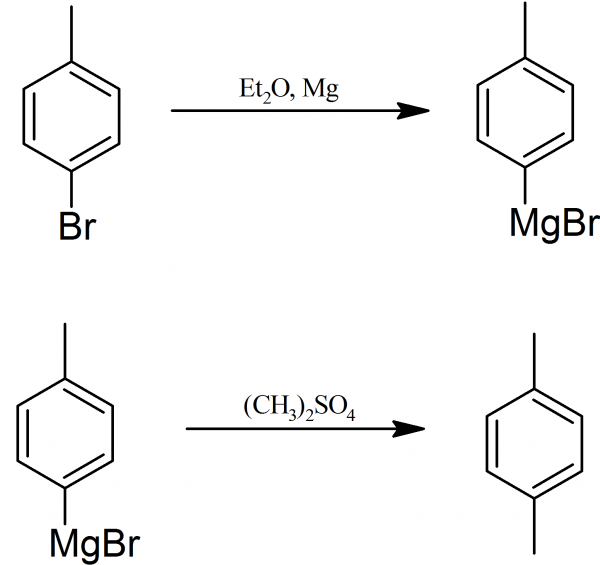


Preparation Of P Xylene
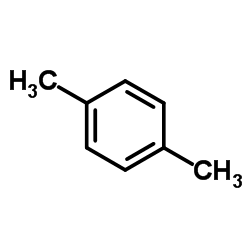


P Xylene Cas 106 42 3 Chemsrc
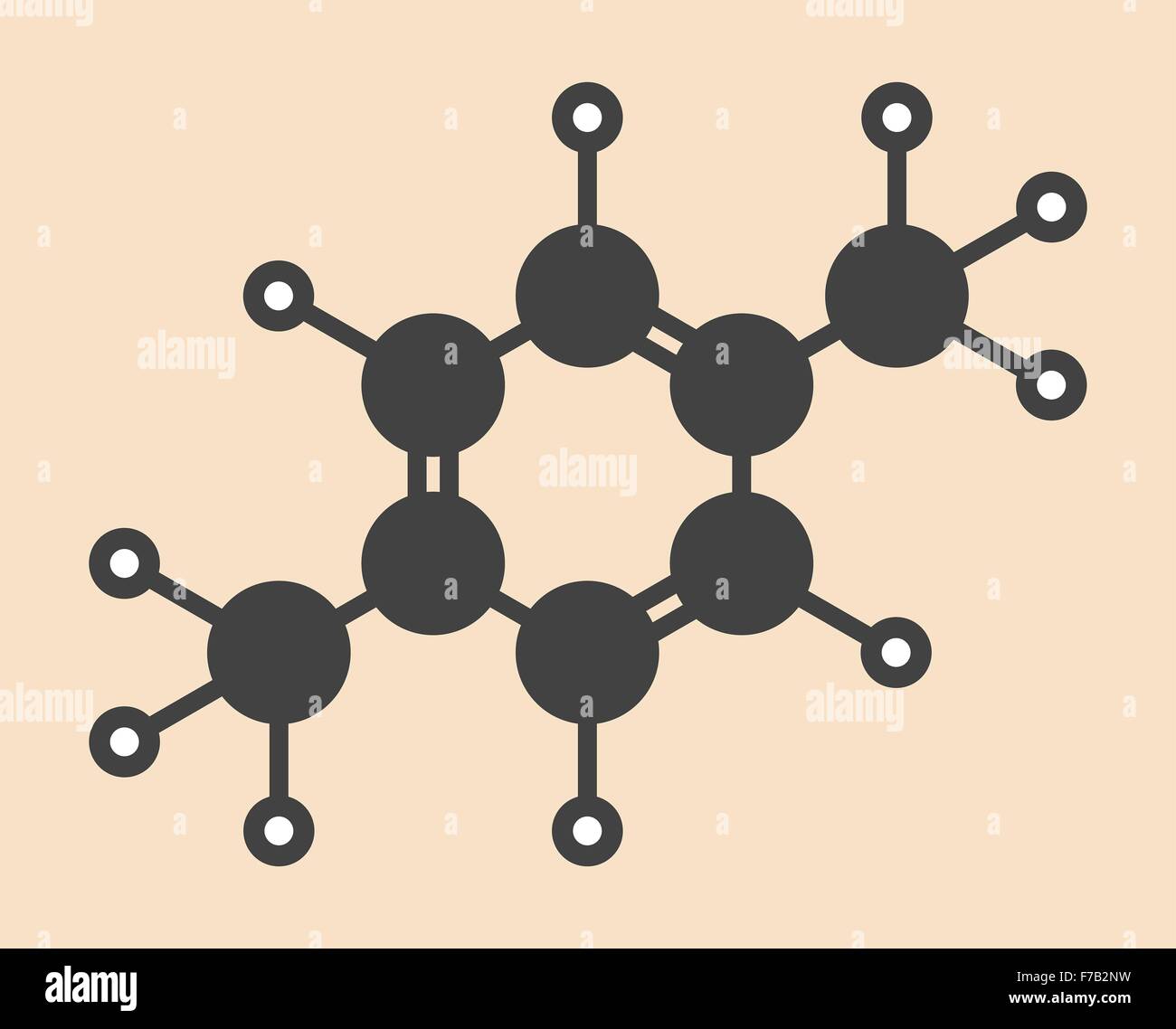


Para Xylene P Xylene Aromatic Hydrocarbon Molecule Stylized Stock Photo Alamy



Difference Between O Xylene And P Xylene Compare The Difference Between Similar Terms
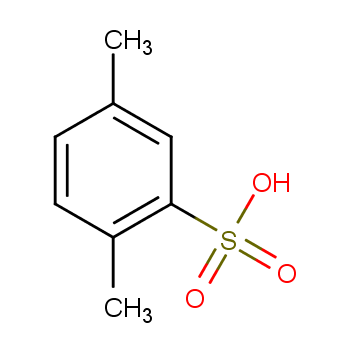


P Xylene 2 Sulphonic Acid 609 54 1 Wiki



Para Xylene P Xylene Molecule Structural Chemical Formula Royalty Free Cliparts Vectors And Stock Illustration Image



The Crystal Structures Of M Xylene And P Xylene C8d10 At 4 5 K Sciencedirect



P Xylene Anhydrous 99 106 42 3 Sigma Aldrich



Selective Methylation Of Toluene Using Co2 And H2 To Para Xylene Science Advances



P Xylene Wikipedia


Q Tbn And9gcsibzrzn0yi2zy 7wkgwhm8epqqvhxojgqxgpzk4qt5euwnuv0j Usqp Cau



Para Xylene An Overview Sciencedirect Topics



Which Of The Given Structure Corresponds To P Xylene


Bond Structure And Polarity P Xylene



P Xylene D10 D 99atom 1 Sigma Aldrich



P Xylene D6 For Nmr 99 Atom D Acros Organics Fisher Scientific



Pxylene Molecular Structure Isolated On Grey Stock Photo Download Image Now Istock



File P Xylene Thiele Skeleton With Conventional Numbering Of Substituent Positions 1 6 Jpg Wikimedia Commons
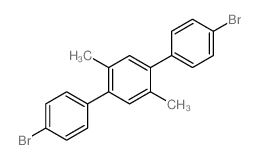


2 5 Bis 4 Bromophenyl P Xylene Cas 55 6 Chemsrc



Alpha Alpha Dibromo P Xylene 97 Acros Organics Fisher Scientific



Solved Draw The Structure Of P Xylene Explain Why The C Chegg Com



Alpha Alpha Alpha Alpha Alpha Alpha Hexachloro P Xylene 98 0 Tci America Fisher Scientific


P Xylene Kovats Retention Index


Lewis Structure



P Xylene 2 Sulfonyl Chloride 98 0 Tci America Fisher Scientific



The Chemical Structure Of O Xylene M Xylene P Xylene And Ethylbenzene Download Scientific Diagram



Xylene Wikipedia


P Xylene C8h10 Chemspider



Raman Spectrum Of P Xylene From 80 30 Cm 1 Under Ambient Conditions Download Scientific Diagram



Science Source Stock Photos Video Ball And Stick Model Of P Xylene
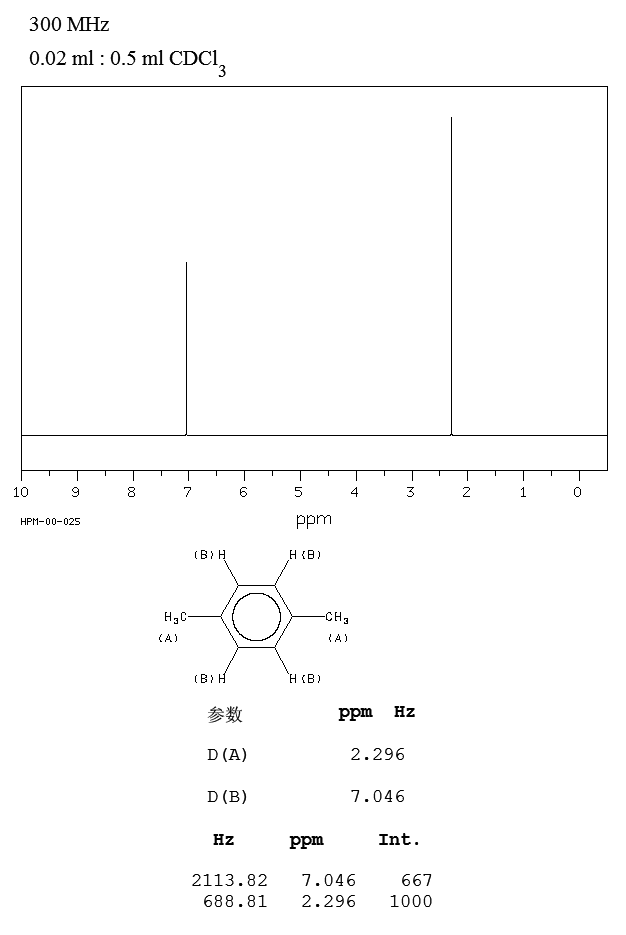


P Xylene 106 42 3 1h Nmr



Discrimination Between O Xylene M Xylene P Xylene And Ethylbenzene By Host Compound R R 2 3 Dimethoxy 1 1 4 4 Tetraphenylbutane 1 4 Diol Sciencedirect


Q Tbn And9gcqboqag54ofdmtxd4xx4eljivqhwf5fgb0okk1cops Usqp Cau



1 2 Xylene 1 3 Xylene 1 4 Xylene C24h30 Pubchem


2 5 Diisopropyl P Xylene C14h22 Pubchem



P Xylene Wikipedia


Q Tbn And9gcsibzrzn0yi2zy 7wkgwhm8epqqvhxojgqxgpzk4qt5euwnuv0j Usqp Cau


2 5 Dichloro P Xylene C8h8cl2 Pubchem



The Chemical Structure Of O Xylene M Xylene P Xylene And Ethylbenzene Download Scientific Diagram
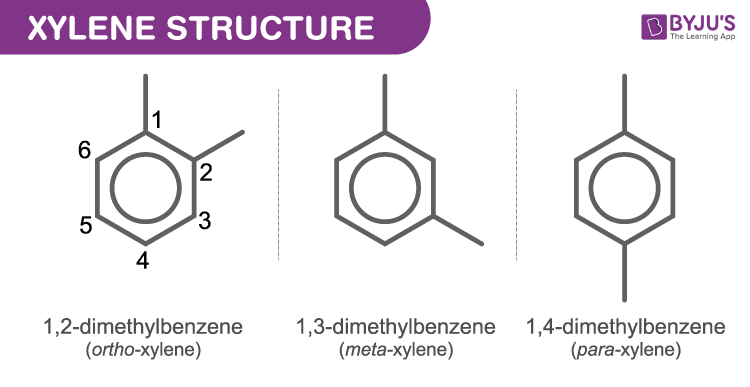


Xylene Properties Structure And Uses Of C8h10


コメント
コメントを投稿